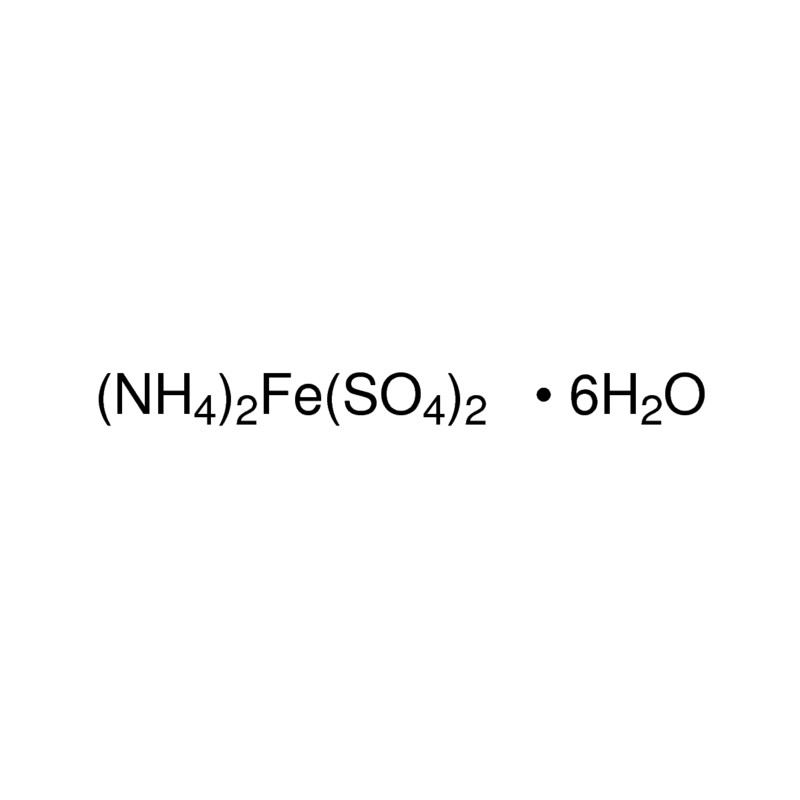When 3.92 g L ^{-1} of sample of Mohr's salt reacts completely with 50 mL N/10 KMnO_4 solution. The percentage purity of the sample of Mohr's salt is :

Fram M HR maut (Mohr's salt) o 0 HGG À KMnO4 in Eth a wiara 3174147 fafe À ptra. 10 Determine the molarity of the given KMnO, solution with the of supplied

The molecular formula of Mohr's salt is (NH.),SO.FeS0.6H,O(1) Find the number of atoms of each element.(2) - Brainly.in

Oxidation state of fe in mohr's salt with solution and formula Step wise explanation needed - Chemistry - Redox Reactions - 16519045 | Meritnation.com

does mohr's salt contain iron ? write the chemical formula of mohr's salt? from brainly - Brainly.in
4 mole of a mixture of Mohr's salt and fe2(so4)3 requires 500 ml of 1 M K2cr2o7 for complete oxidation in acidic media um.mole

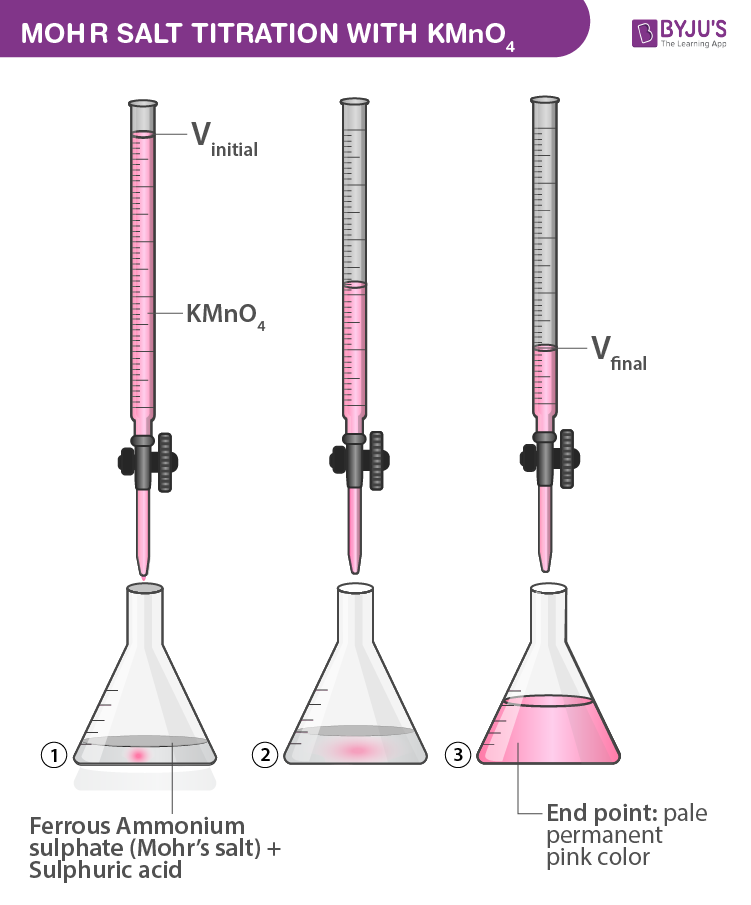

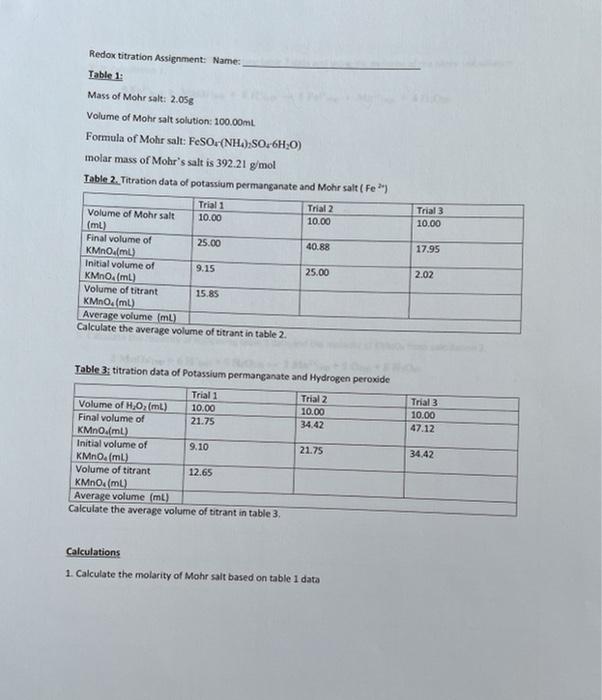




![Odia] What is formula of Mohr's salt. Odia] What is formula of Mohr's salt.](https://static.doubtnut.com/ss/web/11457989.webp)